Class 12 Chemistry - Chapter The Solid State NCERT Solutions | What is a semiconductor? Describe the tw
What is a semiconductor? Describe the two main types of semiconductors and contrast their conduction mechanism.
Semi- conductors are the solids that have properties intermediate between metals and insulators.They have only small difference in energy between the filled valence band and empty conduction band.The conductivity is also intermediate between that of a metal and an insulator and depends upon the number of electrons in the conduction band.
The semiconductors behaves in two types as
a) Intrinsic conduction
b) Extrinsic conduction
(a) Intrinsic conduction = pure silicon and germanium are poor conductors because they have network of four strong covalent bonds. But when electricity is provided to the crystals, the electrons moves leaving behind a positive charge or hole at the site of missing electron, as a result of which the crystal will now conduct electricity.
(b) Extrinsic conduction = here the conductivity of intrinsic conductors is increased by adding an appropriate amount of suitable impurity. This process is called doping. The impurities are of 2 types:
(1) Electron rich
(2) Electron deficient
(1) Electron rich impurity – here atoms with five valence electrons are added to the semiconductor, as a result of which four out of five electrons are used in formation of four covalent bonds with four neighbouring silicon atoms. The fifth electron is extra and becomes delocalised. These delocalised electrons increase the conductivity of doped silicon, hence increases the conductivity. This type of conductor is called n-type semiconductor.
(2) Electron deficient impurity – here an atom with three valence electrons are added to the semiconductor, as a result of which silicon or germanium atom are replaced by the impurity atom. The valence electrons of impurity will make bond with three atom but fourth atom remains free in the crystal of silicon or germanium, which is now available for conducting electricity. This type of conductor is called p-type semi- conductor.
More Questions From Class 12 Chemistry - Chapter The Solid State
- Q:-
If NaCl is doped with 10-3mol % of SrCl2, what is the concentration of cation vacancies?
- Q:-
A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What are the coordination numbers of P and Q?
- Q:-
An element with molar mass 2.7 x 10-2kg mol-1 forms a cubic unit cell with edge length 405 pm. If its density is 2.7 x 103 kg m-3, what is the nature of the cubic unit cell?
- Q:-
Copper crystallises into a fcc lattice with edge length 3.61 x 10-8cm. Show that the calculated density is in agreement with its measured value of 8.92 g cm-3.
- Q:-
A compound is formed by two elements M and N. The element N forms ccp and atoms of M occupy 1/3rd of tetrahedral voids. What is the formula of the compound?
- Q:-
Aluminium crystallises in a cubic close-packed structure. Its metallic radius is 125 pm.
(i) What is the length of the side of the unit cell?
(ii) How many unit cells are there in 1.00 cm3of aluminium?
- Q:-
Analysis shows that nickel oxide has the formula Ni0.98 O1.00. What fractions of nickel exist as Ni2+and Ni3+ions?
- Q:-
Distinguish between
(i)Hexagonal and monoclinic unit cells
(ii) Face-centred and end-centred unit cells.
- Q:-
Solid A is a very hard electrical insulator in solid as well as in molten state and melts at extremely high temperature. What type of solid is it?
- Q:-
Ionic solids, which have anionic vacancies due to metal excess defect, develop colour. Explain with the help of a suitable example.
Popular Questions of Class 12 Chemistry
- Q:-
For the reaction R → P, the concentration of a reactant changes from 0.03 M to 0.02 M in 25 minutes. Calculate the average rate of reaction using units of time both in minutes and seconds.
- Q:-
Write the formulas for the following coordination compounds:
(i) Tetraamminediaquacobalt (III) chloride
(ii) Potassium tetracyanonickelate(II)
(iii) Tris(ethane-1,2-diamine) chromium(III) chloride
(iv) Amminebromidochloridonitrito-N-platinate(II)
(v) Dichloridobis(ethane-1,2-diamine)platinum(IV) nitrate
(vi) Iron(III) hexacyanoferrate(II)
- Q:-
(i) Write structures of different isomeric amines corresponding to the molecular formula, C4H11N
(ii) Write IUPAC names of all the isomers.
(iii) What type of isomerism is exhibited by different pairs of amines?
- Q:-
Write any two characteristics of Chemisorption.
- Q:-
Write the structures of the following compounds.
(i) α-Methoxypropionaldehyde
(ii) 3-Hydroxybutanal
(iii) 2-Hydroxycyclopentane carbaldehyde
(iv) 4-Oxopentanal
(v) Di-sec-butyl ketone
(vi) 4-Fluoroacetophenone
- Q:-
Which of the ores mentioned in Table 6.1 can be concentrated by magnetic separation method?
- Q:-
Why are pentahalides more covalent than trihalides?
- Q:-
Silver atom has completely filled d orbitals (4d10) in its ground state. How can you say that it is a transition element?
- Q:-
Glucose or sucrose are soluble in water but cyclohexane or benzene (simple six membered ring compounds) are insoluble in water. Explain.
- Q:-
Write structures of the following compounds:
(i) 2-Chloro-3-methylpentane
(ii) 1-Chloro-4-ethylcyclohexane
(iii) 4-tert. Butyl-3-iodoheptane
(iv) 1,4-Dibromobut-2-ene
(v) 1-Bromo-4-sec. butyl-2-methylbenzene
Recently Viewed Questions of Class 12 Chemistry
- Q:-
Complete each synthesis by giving missing starting material, reagent or products
(i)
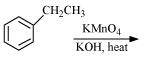
(ii)

(iii)

(iv)

(v)

(vi)
- Q:-
Name a substance which can be used as an antiseptic as well as disinfectant.
- Q:-
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is
(i) doubled
(ii) reduced to half?
- Q:-
In the series Sc (Z = 21) to Zn (Z = 30), the enthalpy of atomization of zinc is the lowest, i.e., 126 kJ mol−1. Why?
- Q:-
Write the mechanism of hydration of ethene to yield ethanol.
- Q:-
Boiling point of water at 750 mm Hg is 99.63°C. How much sucrose is to be added to 500 g of water such that it boils at 100°C.Molal elevation constant for water is 0.52 K kg mol-1.
- Q:-
With what neutral molecule is ClO- isoelectronic? Is that molecule a Lewis base?
- Q:-
Draw all the isomers (geometrical and optical) of:
(i) [CoCl2(en)2]+
(ii) [Co(NH3)Cl(en)2]2+
(iii) [Co(NH3)2Cl2(en)]+
- Q:-
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?

- Q:-
The value of ΔfGø for formation of Cr2O3 is - 540 kJmol-1 and that of Al2 O3 is - 827 kJmol-1. Is the reduction of Cr2O3 possible with Al?
- All Chapters Of Class 12 Chemistry
- All Subjects Of Class 12
