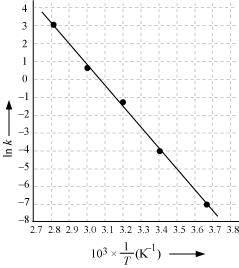
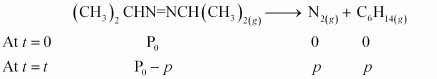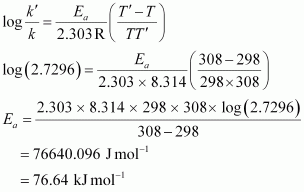Active and Passive Voice Rules
Voice is a crucial aspect of English grammar. It indicates if the subject of the sentence is performing the action or if the action is performed on the subject. To know the difference between the active and passive voice, contributing to the improvement of writing, speaking, and sentence clarity. In this article, you will learn … Read more
Read More